Safe effect of shock wave therapy for Plantar Fascitis
- Type: Free
Radial Extracorporeal Shock Wave TherapyIs Safe and Effective in the Treatment ofChronic Recalcitrant Plantar Fasciitis
Results of a Confirmatory Randomized Placebo-Controlled Multicenter Study
Ludger Gerdesmeyer,*†‡ MD, PhD, Carol Frey,§ MD, Johannes Vester,|| PhD, Markus Maier,¶ PhD, Lowell Weil Jr,# DPM, Lowell Weil Sr,# DPM, Martin Russlies,** PhD, John Stienstra,†† DPM, Barry Scurran,†† DPM, Keith Fedder,§ MD, Peter Diehl,‡‡ MD, Heinz Lohrer,§§ MD, Mark Henne,† MD, and Hans Gollwitzer,† MD From the †Department of Orthopedic and Traumatology, Technical University Munich, Klinikum Rechts der Isar, Germany, the ‡Department of Joint Arthroplasty and Clinical Science, Mare Clinic, Kiel, Germany, §Orthopedic Foot and Ankle Center, Manhattan Beach, California, ||IDV Data Analyses and Study Planning, Biometrics in Medicine, Gauting, Germany, the ¶Department of Orthopedics, Ludwig Maximilian University, Munich, Germany, the #Weil Foot and Ankle Institute, Des Plaines, Illinois, **University Schleswig Holstein, Campus Lübeck, Lübeck, Germany, the ††Department of Podiatry, The Permanente Medical Group Inc, Union City, California, the ‡‡Department of Orthopedics, University Rostock, Rostock, Germany, and the §§Institute of Sports medicine, Frankfurt am Main, Germany |
|
Background: Radial extracorporeal shock wave therapy is an effective treatment for chronic plantar fasciitis that can be administered to outpatients without anesthesia but has not yet been evaluated in controlled trials.
Hypothesis: There is no difference in effectiveness between radial extracorporeal shock wave therapy and placebo in the treatment of chronic plantar fasciitis.
Study Design: Randomized, controlled trial; Level of evidence, 1.
Methods: Three interventions of radial extracorporeal shock wave therapy (0.16 mJ/mm2; 2000 impulses) compared with placebo were studied in 245 patients with chronic plantar fasciitis. Primary endpoints were changes in visual analog scale composite score from baseline to 12 weeks’ follow-up, overall success rates, and success rates of the single visual analog scale scores (heel pain at first steps in the morning, during daily activities, during standardized pressure force). Secondary endpoints were single changes in visual analog scale scores, success rates, Roles and Maudsley score, SF-36, and patients’ and investigators’ global judgment of effectiveness 12 weeks and 12 months after extracorporeal shock wave therapy.
Results: Radial extracorporeal shock wave therapy proved significantly superior to placebo with a reduction of the visual analog scale composite score of 72.1% compared with 44.7% (P = .0220), and an overall success rate of 61.0% compared with 42.2% in the placebo group (P = .0020) at 12 weeks. Superiority was even more pronounced at 12 months, and all secondary Out ome measures supported radial extracorporeal shock wave therapy to be significantly superior to placebo (P < .025, 1- sided). No relevant side effects were observed.
Conclusion: Radial extracorporeal shock wave therapy significantly improves pain, function, and quality of life compared with placebo in patients with recalcitrant plantar fasciitis.
Keywords: heel pain; plantar fasciitis; shock wave; lithotripsy; radial extracorporeal shock wave therapy |
Plantar fasciitis is the most common cause of heel pain and accounts for approximately 11% to 15% of all foot symptoms requiring professional care in the adult.1,4,28 The course of the disease is typically self-limiting, and about 90% of patients are successfully treated with nonsurgical measures.1,2,4 The self-limiting character of the disease also explains the relatively high success rates observed in the placebo arm of double-blind, randomized, controlled trials.1,2,4,8,25,28,38 Nevertheless, the remaining patients enter a state of recalcitrant painful heel syndrome, often requiring operative intervention.1,2,4,8 Thereby, operative treatments like fasciotomy have shown promising results but are often associated with long recovery times, and ath letes especially seek alternative treatment modalities that allow for continued training.1,2,4,8
Extracorporeal shock wave therapy (ESWT) has been introduced for the treatment of recalcitrant painful heel syndrome as an alternative to surgery, allowing fast recovery times without the necessity of reduced weight bearing or immobilization.22,23,28,33,38 However, randomized, controlled trials assessing ESWT in chronic painful heel syndrome have revealed contradictory results, and the clinical effectiveness has been discussed controversially.3-5,8,14,17,18,23,32 By reviewing the published trials it becomes obvious that the different treatment parameters of ESWT are of utmost importance for the outcome of treatment.5,17,22,23,28 In this respect, especially the application of local anesthesia has been shown to reduce efficacy.19,30 Furthermore, higher energies have been associated with greater pain reduction.23,27,33
It becomes apparent that pooling data of different treatment protocols in meta-analyses or systematic reviews is critical.22,28,35 Therefore, in assessing the effectiveness of ESWT in plantar fasciitis, only specific treatment protocols should be evaluated, and results could not be generalized.
Radial ESWT (rESWT) has been introduced into medicine as an effective and easy method to apply shock wave technology.15,20It represents an alternative to focused shock wave treatment, allowing for a broader application. Radial shock waves are generated ballistic ally by accelerating a bullet to hit an applicator, which transforms the kinetic energy into radially expanding shock waves.15,20 Compared with these radial shock waves, the focused shock waves show deeper tis- sue penetration with significantly higher energies concentrated to a smaller focus.15,16,20,22,28 This article reports on a randomized, controlled, and double-blinded Food and Drug Administration (FDA) study evaluating the efficacy and safety of rESWT in patients with chronic painful heel syndrome.
METHODS
Study Design and Follow-up
This double-blind, randomized, placebo-controlled trial with parallel group design was conducted internationally at 3
Study centers in the United States and 5 study centers in Europe. Patient enrollment took place during an 11-month period. A total of 254 patients were randomly assigned to receive either rESWT or placebo treatment with concealed allocation in permuted blocks of 4 to 8, stratified by treatment center with the use of a computer-generated random list (Rancode; idv Data Analysis and Study Planning, Gauting, Germany). Concealment of randomization was guaranteed by nontransparent envelopes. Both patients and assessing physicians were blinded to randomization as well as to the evaluating physician. The trial was conducted as an FDA approval study. In designing the study, the authors adhered to the standardized guidelines of good clinical practice from the International Conference on Harmonization (ICH).11,12
After 3 shock wave or placebo interventions were applied, patients were followed until the end of the follow-up 1 period (12 weeks after the last intervention). At this visit, the participants’ response to treatment was rated, and patients who showed sufficient response on a clinically relevant level continued the follow-up 2 phase, which ended 12 months after the last rESWT or placebo intervention. If patients suffered from significant pain after intervention, deblinding on demand after 12 weeks was allowed to provide other treatment options outside the trial instead of suffering for 12 months. These patients left the trial with the worst outcome, which was carried forward. All these “worst” data were carried for- ward and used for analysis.
Participants
Patients were recruited from the participating study sites and from community-based referring physicians (primary care physicians, podiatrists, orthopedic surgeons). Participants of all activity levels were included and were asked to continue on the same activity level throughout the study, although activity level at enrollment and during the study were not specifically assessed. The study was approved by the FDA and the responsible independent institutional review boards. Written informed consent was obtained from all participants. A total of 495 patients with plantar heel pain were screened; 254 patients met enrollment criteria and were enrolled in the study. A total of 251 patients out of 254 were treated. The flow of participants through the study is displayed in Figure 1.
Inclusion Criteria
The complete inclusion criteria are listed in Appendix A (available online at http://ajs.sagepub.com/supplemental/). Inclusion criteria included a history of at least 6 months of chronic plantar painful heel syndrome that proved resist- ant to nonsurgical treatment. Diagnosis was confirmed clinically by physical examination with a typical point of
Study centers in the United States and 5 study centers in Europe. Patient enrollment took place during an 11-month period. A total of 254 patients were randomly assigned to receive either rESWT or placebo treatment with concealed allocation in permuted blocks of 4 to 8, stratified by treatment center with the use of a computer-generated random list (Rancode; idv Data Analysis and Study Planning, Gauting, Germany). Concealment of randomization was guaranteed by nontransparent envelopes. Both patients and assessing physicians were blinded to randomization as well as to the evaluating physician. The trial was conducted as an FDA approval study. In designing the study, the authors adhered to the standardized guidelines of good clinical practice from the International Conference on Harmonization (ICH).11,12
After 3 shock wave or placebo interventions were applied, patients were followed until the end of the follow-up 1 period (12 weeks after the last intervention). At this visit, the participants’ response to treatment was rated, and patients who showed sufficient response on a clinically relevant level continued the follow-up 2 phase, which ended 12 months after the last rESWT or placebo intervention. If patients suffered from significant pain after intervention, deblinding on demand after 12 weeks was allowed to provide other treatment options outside the trial instead of suffering for 12 months. These patients left the trial with the worst outcome, which was carried forward. All these “worst” data were carried for- ward and used for analysis.

Figure 1. Flow of participants through the study.
aSafety population: all patients receiving at least one treatment session.
bITT population, intent-to-treat. All patients who had at least one treatment session and also at least one evaluation after the first treatment without severe deviation of entry criteria “full analysis set,” according to ICH E9 Biostatistics.11,12
cPP population, per protocol. Exclusion of patients from ITT population with protocol violations (inclusion/exclusion criteria, incomplete study treatment, premature discontinuation).
dSufficient response was considered at least a 60% reduction in pain on 2 of the 3 VAS scales (overall success VAS), or if less pain reduction, then the patient had to be able to work and complete activities of daily living, had to be satisfied with the outcome of the treatment, and must not have required any other treatment to control heel pain. Participants were requested to continue until follow-up 2 (12 months).
eData reflects last value carried forward (LVCF) replacement of missing values of “nonresponders” in the ITT population.
maximum tenderness over the medial tubercle of the calcaneus.1,4 To be eligible, participants had to score significant pain of at least 5 or greater on all 3 visual analog scale (VAS) scores (with a maximum of 10), must have had significant limitation on the Roles and Maudsley Score (fair or poor), and had to have failed results from at least 2 non pharmacological and 2 pharmacological treatments. All patients had to respect a sufficient washout period after each intervention prior to enrollment. The specific washout phases were determined as at least 6 weeks from last corticosteroid injection; 4 weeks from the last local anesthetic injection, iontophoresis, ultrasound, and electrotherapy; 1 week from the last intake of no steroidal antiinflamma-tory drugs (NSAIDs); and 2 days from last heat, ice, mas- sage, stretching, or modification of night splinting and orthotics.
Exclusion Criteria
Reasons for exclusion are listed in the Appendix B (avail- able online at http://ajs.sagepub.com/supplemental/). Major reasons for exclusion were rheumatic or other systemic inflammatory disease, osteomyelitis, active infection or his- tory of chronic infection in the treatment area, neurological or vascular insufficiencies, nerve entrapment syndrome, disturbance of coagulation or ongoing anticoagulatory therapy, significant bilateral heel pain in need of medical treatment, and pregnancy.
Study Procedures
Randomization and treatment were started within 28 days after screening. Radial ESWT or identical placebo were administered in 3 sessions, each 2 weeks (±4 days) apart. A total of 2000 shock waves of the assigned intervention were delivered per session with the Swiss Doloclast radial shock wave device (EMS Electro Medical Systems, Nyon, Switzerland). Before the intervention, the point of maxi- mum tenderness was clinically located by the treating clinician, and the hand-piece was coupled to the identified area by using specific ultrasound coupling gel (EMS Electro Medical Systems).
In the treatment group, 2000 impulses of radial shock waves with an energy flux density of 0.16 mJ/mm2 and a rate of 8 impulses per second were applied at each treatment session. Patients in the control group received identical placebo intervention with a placebo hand-piece that prevented transmission of shock waves. The placebo hand- piece was identical in design, shape, and weight to ensure that there was no way to identify the placebo hand-piece. The treatment in the placebo group was the same com- pared with the active one. Thereby, set up and sound created by the shock wave device was identical in both groups; how- ever, no energy was administered in the placebo group. The intervention was performed in the office by the non- blinded orthopedic surgeon or podiatrist, locating the tip of the applicator to the most tender point at the medial calcaneal tubercle, controlling proper placement by patient-controlled feedback, and adjusted during treatment if necessary.
A standardized rescue medication was allowed through- out the entire study if pain became unbearable (2 g of acetaminophen per day for up to 14 days after the last intervention; thereafter, 2 g of acetaminophen per week). No other therapies were allowed, and orthotics could not be modified until the 12-week follow-up (follow-up 1).
Outcome Measures
The primary outcome measure was overall heel pain reduction measured by the percentage change of the VAS composite score 12 weeks after treatment compared with baseline, with last value carried forward (LVCF) replacement of missing values with the last recorded value and correction for interfering analgesic therapy. The 12-month analysis was performed in the same manner by replacing missing values in LVCF and correction for interfering analgesic therapies with the last recorded value. The heel pain composite score was defined as the sum of three 10-cm VAS scores: heel pain when taking the first steps in the morning, heel pain while doing daily activities, and heel pain while applying a standardized local pressure with the Dolor meter (EMS Electro Medical Systems) to quantify local pressure pain. The Dolor meter is a device that allows for objectivity of pressure application, with an integrated scale to exactly determine the applied local pressure.17 The blinded investigator used the Dolor meter to measure pressure sensitivity at the point of maximum tenderness. The pressure level measured at the Dolor meter scale that just elicited unbearable pain was documented as baseline value and was quantified by the Dolor meter on the 10-cm VAS scale. At each follow-up visit, the same Dolor meter pressure was applied, and the subject was asked to score the pain on the VAS. The increased pressure pain tolerance reflects the shock wave–induced effect.
The further primary efficacy criteria were the single success rates and the overall success rate with regard to heel pain, defined as percentage decrease of heel pain larger than 60% from baseline at 12 weeks after treatment for at least 2 of the 3 heel pain (VAS) measurements. To keep the multiple level of α, the further set of primary efficacy criteria were tested in the a priori–ordered sequence according to the principle of a priori–ordered hypotheses.25
The primary endpoint for comparison of groups was 12 weeks after the last treatment. At this point the decision was also made whether the patient had sufficient treatment response to continue the study. Sufficient response was considered a minimum 60% reduction in pain on 2 of 3 VAS scores. Patients who showed sufficient and clinically relevant response to rESWT were to continue in the follow- up 2 period (12 months after last rESWT). Patients who suffered from significant pain after intervention deblinding on demand after 12 weeks were allowed to explore other treatment options outside the trial instead of suffer- ing for 12 months. These patients left the trial with the worst outcome, the data of which were carried forward and used for analysis.
Secondary outcome measures were changes in Roles and Maudsley score, SF-36 physical percent changes, SF-36 Mental percent changes, investigator’s judgment of effectiveness (7-point scale), patient’s judgment of therapy satisfaction (7-point scale), and patient recommendation of therapy to a friend (all at 12 weeks after treatment). Furthermore, VAS composite score, success rates, and single VAS scores were assessed after completion of follow-up 2 (12 months after last treatment session).
Safety Criteria
All patients who had at least one treatment session were analyzed for safety. Patients were followed throughout the study, and all local tissue effects and adverse events were recorded. Additionally, the investigator’s global judgment of tolerability was assessed on a 7-point rating scale 12 weeks after the last treatment. To assess local adverse event tendon rupture observation, Semmes-Weinstein5.07 (10 g) Monofilament Assessment, Toe Clawing Observation, and Ankle-Brachial Assessment of the lower extremity were performed at each visit. All assessments but one were done by clinical examination. The Semmes- Weinstein Monofilament was used to assess global neurological deficiencies. A 10-gram (5.07 log) monofilament wire was applied to each foot at 10 sites. Loss of protective sensation generally is indicated by a patient’s inability to feel the monofilament at 4 or more of the 10 sites.
Statistical Analysis
The study had a statistical power of 90% to detect a reduction by 50 percentage points in the primary outcome measure of a reduction of VAS composite score from baseline to
12 weeks after completion of shock wave treatment. A dropout rate of 10% was calculated as well before study start.
To keep the multiple level of α, the set of primary efficacy criteria were tested in the a priori–ordered sequence according to Maurer.25 By this, if the first test is statistically significant (VAS composite score), the second test (overall success rate) can be performed to confirm with full level of α. If the second test (overall success rate) is also statistically significant, the single success rates can be tested to confirm with full α in the sequence “heel pain when taking first steps of the day,” “heel pain while doing daily activities,” “heel pain after application of the Dolor meter” as long as the preceding test is statistically significant. Efficacy of the rESWT treatment is proved if at least the first hypothesis (VAS composite score) shows a statistically significant result. A value of P < .025 (1-sided) was considered statistically significant.
To identify differences in effect size between the different intervention groups, the Mann-Whitney effect size with predefined benchmarks was used to define the probability that a randomly selected participant from the rESWT group was better off than a randomly selected participant from the control group. In accordance with Colditz et al,6 the authors used relevant benchmarks that corresponded to a Mann-Whitney effect size of 0.5 for equality (active therapy no better or worse than placebo); 0.44 or
0.56 for small-sized inferiority or superiority, respectively;

size demonstrated more than small superiority of rESWT for all 5 a priori–ordered hypotheses compared with placebo (Mann-Whitney effect size >0.56), demonstrating significant as well as clinically relevant effects.
Regarding follow-up 2 (12 months after rESWT) the P values of all 5 primary efficacy criteria were far below the predefined level of significance (.025, 1-sided, exploratory interpretation, Table 2). Nonresponders of follow-up 1 period and premature discontinuations were included by
LVCF replacement of missing values and for safety analysis.
These results even demonstrated a pronounced treatment effect at follow-up 2, and treatment success was maintained. The composite VAS score of the heel pain was –84.8% in the patients treated actively and –43.2% in the placebo group (P < .025). Thus, there is strong evidence for long-term superiority of the ESWT treatment compared with identical placebo.
To assess the stability of the results, different sensitivity analyses were performed for the primary efficacy criteria at follow-up 1 and 2 assessing a per-protocol analysis, a supportive analysis for the ITT data set without any correction for interfering analgesic therapy, a further supportive
sensitivity analysis for the ITT data set with correction for interfering analgesic therapy by means of the worst rank score technique, and an analysis of the data set with the “data as available” instead of LVCF replacement of missing values. All in all, the results of the sensitivity
analyses support the results of the confirmatory analysis (see Appendix D for details, available online at http://ajs.sagepub.com/supplemental/).
Secondary Outcome Measures The efficacy results demonstrated superiority of the rESWT group not only in the confirmatory analyses, but also in the supportive sensitivity analyses and in the analysis of the secondary outcome measures. All tested secondary efficacy criteria,
including the SF-36 score, the Roles and Maudsley Score, global judgment of effectiveness, therapy satisfaction, and therapy recommendation, showed better outcome at the primary endpoint in favor of the rESWT group, and all test results were statistically significant (P < .025 1-sided).
Furthermore, all Mann-Whitney effect sizes denoted more than small superiority of the rESWT group compared with placebo (Table 3). Thus, there is evidence for benefit of patients with painful heel by rESWT treatment not only through reduced pain but also by generally improved wellbeing.
The positive result was also reflected by the high recommendation rate of participants with rESWT treatment with a final recommendation of study therapy in 91.2% of the rESWT group compared to 69.1% of the participants with placebo intervention.
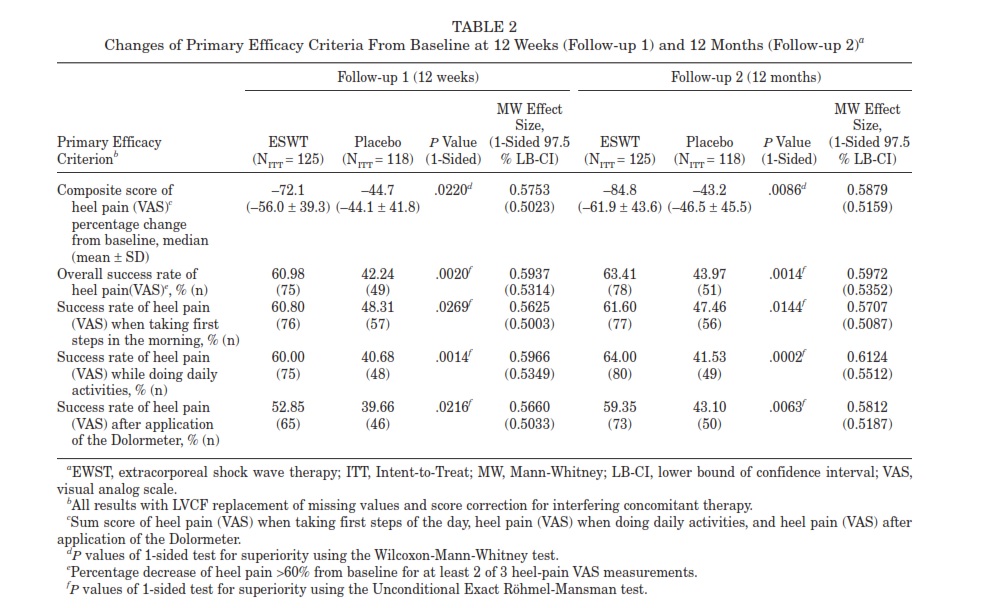
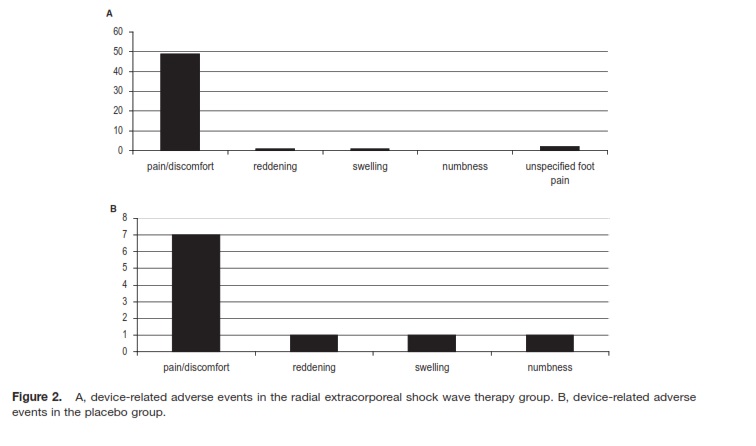
Adverse Events and Safety Criteria A total of 251 subjects received at least one treatment session (“safety population”; Figure 1). There were 50 devicerelated adverse events in 33 patients of the rESWT group and 11 adverse events in 10 subjects in the placebo group.
A total of 46 of 50 device-related nonserious adverse events in the ESWT group was due to pain and discomfort during treatment as displayed in Figure 2. The duration of discomfort was reported as maximal 10 minutes mostly, and no participant requested local anesthesia during rESWT, even though this was offered to all patients. Device-related adverse events had no influence on outcome. No adverse or severe adverse event occurred regarding tendon rupture observation, Semmes-Weinstein 5.07 (10 g) Monofilament Assessment, Toe Clawing Observation, and Ankle-Brachial Assessment.
At the primary endpoint 12 weeks after the last treatment, the tolerability of the treatment was judged by the investigator as “very good” or “good” in 93.8% of the rESWT subjects and in 90.1% of the placebo group.
DISCUSSION
The present placebo-controlled study was conducted to investigate the safety and effectiveness of rESWT in the treatment of recalcitrant plantar fasciitis. Plantar fasciitis is a common complaint and can be especially disabling in
1,4,29,31athletes.Goals of treatment are pain relief and restoration of function. Martin et al 24 and Crawford and 8 Thomson reviewed numerous studies of nonsurgical treatment for plantar fasciitis such as stretching, cryotherapy, heel cushions and shoe inserts, night splints, custom-made orthotics, anti-inflammatory drugs, corticosteroid injection, and immobilization and showed success rates ranging from 44% to 90%.1,4,8,24Nevertheless, not more than limited evidence of efficacy could be demonstrated.
4,8 For patients with chronic heel pain resistant to nonsurgical treatment, surgical interventions are suggested. Surgery, however, can be associated with prolonged healing 8,9 and did not prove superior to ESWT.
9,38 Extracorporeal shock wave therapy for plantar fasciitis has been investigated in multiple well-designed, randomized, and placebo-controlled trials, providing evidence of effectiveness and safety of treatment. 17,23,27,29,30,31,33 These studies also demonstrated significant influence of treatment regimen and concomitant anesthesia on outcome.
19,30 Notably, some double-blind randomized, controlled trials that failed to show the superiority of ESWT over placebo focused the acoustic energy at anatomical landmarks rather than at the point of greatest tenderness as defined by the participant, and local anesthesia was used in some of the investigations in an effort to blind the participants.
5,18,32Recent studies have demonstrated that localanesthesia may inhibit direct analgesic effects like themodification of the release of pain mediators, hyperstimulation,and the gate-control mechanism.
18,21,22,28,37Theseobservations were further supported by a study by Rompeet al 30demonstrating ESWT applied without local anesthesia to be significantly more effective than ESWT used with local anesthesia.
Compared with focused shock wave applicators, rESWT devices address larger treatment areas, thus providing potential advantages in superficial applications like tendinopathies and skin conditions.
7,15,16,28 For deep soft tissue treatments or bone injuries, the radial technique has some limitations regarding penetration depth and higher energy levels.
7,15,16In contrast to so-called focused shock wave therapies, the radial technique is used to treat the painful region rather than a painful point. It is well known that heel pain originates from a painful area along the plantar fascia rather than a certain locally limited spot. From the technical point of view, radial shock wave therapy addresses more the area of pathologic changes compared with focused devices.
10,15,22,30 Due to the patient controlled application and the missing need to control the
|
Figure 2. A, device-related adverse events in the radial extracorporeal shock wave therapy group. B, device-related adverse events in the placebo group. |
focus zone, radial shock wave therapy seems to be easier to apply. 15,20 Because treatment regimen and shock wave parameters have significant influence on outcome, pooling of data in systematic reviews and meta-analyses is critical, and effectiveness has to be assessed for the different devices and treatment protocols. Buchbinder 5,17,27,32,33,35 5 et al 18 and Haake et al used local anesthesia or nerve blocks and found no difference. When no local anesthesia
was used, Gollwitzer et al,17 Rompe et al,30 and Malay et al23 found significantly better outcome after shock wave treatment. In the present study, radial shock waves were oriented without anesthesia by patient-guided feedback to the point of maximum tenderness.
The present randomized, placebo-controlled study demonstrated significant improvement of pain scales and functional measurements, as well as quality of life, after rESWT at follow-up compared to baseline. Furthermore, rESWT proved superior to placebo with regard to the primary outcome measures of “changes in VAS composite score of heel pain” and “overall success rate” at 12 weeks and all secondary outcome measures at 12 months after intervention. At the time of follow-up 1, the VAS composite score was reduced by 72.1% in the rESWT group compared with 44.7% in the placebo group, which was statistically significant. Furthermore, the between-group difference of nearly 30% is considered clinically relevant.13 The superiority of rESWT compared with placebo was not only limited to the primary criterion of effectiveness but also strongly sup- ported by the results of the outcome measures’ overall success rate, success rate of heel pain while doing daily activities, success rate of heel pain after the application of the Dolor meter, Roles and Maudsley score, percentage changes from baseline of the SF-36 summary measures mental and physical health, investigator’s and patient’s judgment of effectiveness, and the patient’s rate of recommendation of the applied therapy to a friend. An additional assessment of the Mann-Whitney effect size revealed more than small superiority of rESWT in all determined criteria both at 12 weeks and at 12 months after the intervention. Furthermore, several sensitivity analyses were performed to check the stability of the data, which all corroborate effectiveness of rESWT compared with placebo showing significant superiority (see Appendix D, available online at http://ajs.sagepub.com/ supplemental/).
Regarding the second follow-up at 12 months after shock wave treatment, superiority of rESWT compared with placebo was even more pronounced, with a reduction of the VAS composite score from baseline of 84.8% after shock wave treatment compared with 43.2% in the placebo group, demonstrating a more than 40% between-group difference. All other outcome measures also showed superiority of the rESWT group, thus proving excellent long-term efficacy and supporting the application of rESWT in the treatment of chronic plantar fasciitis. Apart from the investigated pain scales, we were able to demonstrate significant improvement in the scores evaluating quality of life (SF-36) and function (Roles and Maudsley score). Thus, there is evidence for benefit of patients with painful heel by ESWT treatment not only through reduced pain but also by generally improved well-being.
Treatment was applied without anesthesia, and our results demonstrated rESWT to be safe with excellent tolerability. The results demonstrate that patients assigned the rESWT were found to have more pain during treat- ment compared with placebo but no one required any local anesthetic. The sensitivity analysis demonstrates no differences in outcome with regard to all side effects.
From the clinical point of view the outcome regarding recommendation and global judgment by patients and investigators demonstrated high clinical impact and acceptance. Notably, nearly 70% of patients of the placebo group would also recommend the received treatment to a friend. This large placebo effect demonstrates the effectiveness of the blinding technique, which also was found in other randomized, controlled studies.17,23,35 In review, with a mean improvement of the VAS composite score of more than 40% in the placebo group at follow-up compared with baseline, the power of the placebo effect in these kinds of studies becomes obvious and has to be addressed by randomized, placebo-controlled studies to analyze the pure placebo effect. The associated placebo effect is related to the device itself but also to the procedure and the physician.26,34,36 The pure treatment effect and the associated so- called placebo effect are not distinguishable and clinically used together.26,34,36
Radial ESWT demonstrated safety and effectiveness with a protocol of 3 consecutive treatments (3 × 2000 impulses, 0.16 mJ/mm2), applied without anesthesia to the spot of greatest tenderness. Radial ESWT can be strongly recommended for patients with therapy-resistant plantar painful heel syndrome. Especially in the cases of failed nonsurgical treatment, rESWT represents an excellent alternative to surgery because anesthesia is not required and long recovery times are avoided. In addition, partici- pants were not required to refrain from any sports activities during the course of the study. Furthermore, rESWT represents an effective treatment modality that can be administered on an outpatient basis.
ACKNOWLEDGMENT
The study was supported by Electro Medical Systems, Nyon, Switzerland. The sponsor of the study did not have any influence on data collection, analysis, or publication. No constraints were placed on publication of the data.
REFERENCES
- ACFAS Clinical Practice Guideline Heel Pain Panel. The diagnosis and treatment of heel pain. J Foot Ankle Surg. 2001;40:329-340.
- 2. Atkins D, Crawford F, Edwards J, Lambert M. A systematic review of treatments for the painful heel. Rheumatology (Oxford). 1999;38:968-973.
- 3. Buch M, Knorr U, Fleming L, et al. Extracorporeal shockwave therapy in symptomatic heel spurs: an overview. Orthopade. 2002;31:637-644.
- Buchbinder R. Clinical practice. Plantar fasciitis. N Engl J Med.
2004;350:2159-2166.
- 5. Buchbinder R, Ptasznik R, Gordon J, Buchanan J, Prabaharan V, Forbes A. Ultrasound-guided extracorporeal shock wave therapy for plantar fasciitis: a randomized, controlled trial. JAMA. 2002;288:1364-1372.
- Colditz GA, Miller JN, Mosteller F. Measuring gain in the evaluation of medical technology: the probability of a better outcome. Int J Technol Assess Health Care. 1988;4:637-642.
- Coleman AJ, Choi MJ, Saunders JE. Theoretical predictions of the acoustic pressure generated by a shock wave lithotripter. Ultrasound Med Biol. 1991;17:245-255.
- Crawford F, Thomson C. Interventions for treating plantar heel pain.
Cochrane Database Syst Rev. 2003;CD000416.
- Davies MS, Weiss GA, Saxby TS. Plantar fasciitis: how successful is surgical intervention? Foot Ankle Int. 1999;20:803-807.
- Eisenmenger W, Du XX, Tang C, et al. The first clinical results of
“wide-focus and low-pressure” ESWL. Ultrasound Med Biol.
2002;28:769-774.
- 11. European Medicines Agency. ICH Topic E6, Guideline for Good Clinical Practice. http://www.emea.europa.eu/pdfs/human/ich/013595en.pdf. Published July 2002; accessed August 14, 2008.
- 12. European Medicines Agency. ICH Topic E9, Statistical Principles for
Clinical Trials. http://www.emea.europa.eu/pdfs/human/ich/036396en
.pdf. Published September 1998; accessed August 14, 2008.
- Farrar JT, Young JP Jr, LaMoreaux L, Werth JL, Poole RM. Clinical importance of changes in chronic pain intensity measured on an 11- point numerical pain rating scale. Pain. 2001;94:149-158.
- Furia JP. The safety and efficacy of high-energy extracorporeal shock wave therapy in active, moderately active, and sedentary patients with chronic plantar fasciitis. Orthopedics. 2005;28:685-692.
- Gerdesmeyer L, Maier M, Haake M, Schmitz C. Physical-technical principles of extracorporeal shockwave therapy (ESWT). Orthopade.
2002;31:610-617.
- 16. Gerdesmeyer L, Schrabler S, Mittelmeier W, Rechl H. Tissue-induced changes of the extracorporeal shockwave. Orthopade. 2002;31:618-622.
- 17. Gollwitzer H, Diehl P, von Korff A, Rahlfs VW, Gerdesmeyer L.
Extracorporeal shock wave therapy for chronic painful heel syndrome: a prospective, double-blind, randomized trial assessing the efficacy of a new electromagnetic shock wave device. J Foot Ankle Surg. 2007;
46:348-357.
- Haake M, Buch M, Schoellner C, et al. Extracorporeal shock wave therapy for plantar fasciitis: randomised, controlled, multicentre trial. BMJ. 2003;327:75.
- Labek G, Auersperg V, Ziernhold M, Poulios N, Bohler N. Influence of local anesthesia and energy level on the clinical outcome of extracor- poreal shock wave-treatment of chronic plantar fasciitis. Z Orthop Ihre Grenzgeb. 2005;143:240-246.
- Magosch P, Lichtenberg S, Habermeyer P. Radial shock wave therapy in calcifying tendinitis of the rotator cuff—a prospective study. Z Orthop Ihre Grenzgeb. 2003;141:629-636.
- Maier M, Averbeck B, Milz S, Refior HJ, Schmitz C. Substance P and prostaglandin E2 release after shock wave application to the rabbit femur. Clin Orthop Relat Res. 2003;406:237-245.
- Maier M, Milz S, Wirtz DC, Rompe JD, Schmitz C. Basic research of applying extracorporeal shockwaves on the musculoskeletal system: an assessment of current status. Orthopade. 2002;31:667-677.
- 23. Malay DS, Pressman MM, Assili A, et al. Extracorporeal shockwave therapy versus placebo for the treatment of chronic proximal plantar fasciitis: results of a randomized, placebo-controlled, double- blinded, multicenter, intervention trial. J Foot Ankle Surg.
2006;45:196-210.
- Martin RL, Irrgang JJ, Conti SF. Outcome study of subjects with inser- tional plantar fasciitis. Foot Ankle Int. 1998;19:803-811.
- Maurer W, Hothorn LA, Lehmacher W. Multiple comparisons in drug clinical trials and preclinical assays: a priori–ordered hypotheses. In: Vollmar J, ed. Testing Principles in Clinical and Preclinical Trials. Stuttgart, Germany: Gustav Fischer; 1995:3-18.
- Moseley JB, O’Malley K, Petersen NJ, et al. A controlled trial of arthroscopic surgery for osteoarthritis of the knee. N Engl J Med.
2002;347:81-88.
- 27. Ogden JA. Extracorporeal shock wave therapy for plantar fasciitis: ran- domised, controlled, multicentre trial. Br J Sports Med. 2004;38:382.
- Rompe JD, Buch M, Gerdesmeyer L, Haake M, Loew M, Maier M, Heine J. Musculoskeletal shock wave therapy—current database of clinical research. Z Orthop Ihre Grenzgeb. 2002;140:267-274.
- 29. Rompe JD, Decking J, Schoellner C, Nafe B. Shock wave application for chronic plantar fasciitis in running athletes: a prospective, randomized, placebo-controlled trial. Am J Sports Med. 2003;31:268-275.
- Rompe JD, Meurer A, Nafe B, Hofmann A, Gerdesmeyer L. Repetitive low-energy shock wave application without local anesthesia is more efficient than repetitive low-energy shock wave application with local anesthesia in the treatment of chronic plantar fasciitis. J Orthop Res.
2005;23:931-941.
- Rompe JD, Nafe B, Furia JP, Maffulli N. Eccentric loading, shock- wave treatment, or a wait-and-see policy for tendinopathy of the main body of tendo achillis: a randomized, controlled trial. Am J Sports Med. 2007;35:374-383.
- Speed CA, Nichols D, Wies J, Humphreys H, Richards C, Burnet S, Hazleman BL. Extracorporeal shock wave therapy for plantar fasciitis: a double-blind, randomized, controlled trial. J Orthop Res.
2003;21:937-940.
- Theodore GH, Buch M, Amendola A, Bachmann C, Fleming LL, Zingas C. Extracorporeal shock wave therapy for the treatment of plantar fasciitis. Foot Ankle Int. 2004;25:290-297.
- Thomas JR. Placebo surgery. Mo Med. 1999;96:41.
- Thomson CE, Crawford F, Murray GD. The effectiveness of extra cor- poreal shock wave therapy for plantar heel pain: a systematic review and meta-analysis. BMC Musculoskelet Disord. 2005;6:19.
- Tjomsland O, Ekeberg O, Saatvedt K. Placebo effect in research related to surgery and technical procedures [in Norwegian]. Tidsskr Nor Laegeforen. 2001;121:2290-2293.
- Wang CJ, Huang HY, Pai CH. Shock wave-enhanced neovasculariza- tion at the tendon-bone junction: an experiment in dogs. J Foot Ankle Surg. 2002;41:16-22.
38. Weil LS Jr, Roukis TS, Weil LS, Borrelli AH. Extracorporeal shock wave therapy for the treatment of chronic plantar fasciitis: indications, protocol, intermediate results, and a comparison of results to fas- ciotomy. J Foot Ankle Surg. 2002;41:166-172
 Tecar
Tecar Shock Wave
Shock Wave Plasma
Plasma Laser
Laser Tesla
Tesla Nikola
Nikola Kayser
Kayser Fusion
Fusion