Shock wave therapy induces neovascularization at the tendon-bone junction A study in rabbits
- Type: Free
exonAbstract
Despite the success in clinical application, the exact mechanism of shock wave therapy remains unknown. We hypothesized that shock wave therapy induces the ingrowth of neovascularization and improves blood supply to the tissues. The purpose of this study was to investigate the effect of shock wave therapy on neovascularization at the tendon-bone junction. Fifty New Zealand white rabbits with body weight ranging from 2.5 to 3.5 kg were used in this study. The right limb (the study side) received shock wave therapy to the Achilles tendon near the insertion to bone. The left limb (the control side) received no shock wave therapy. Biopsies of the tendon-bone junction were performed in 0, 1,4, 8 and 12 weeks. The number of neo-vessels was examined microscopically with heniatoxylin-eosin stain. Neovascularization was confirmed by the angiogenic markers including vessel endothelial growth factor (VEGF) and endothelial nitric oxide synthase (eNOS) expressions and endothelial cell proliferation determined by proliferating cell nuclear antigen (PCNA) expression examined microscopically with immunohistochemical stains. The results showed that shock wave therapy produced a significantly higher number of neo-vessels and angiogenesis-related markers including eNOS, VEGF and PCNA than the control without shock wave treatment. The eNOS and VEGF began to rise in as early as one week and remained high for 8 weeks, then declined at 12 weeks; whereas the increases of PCNA and neo-vessels began at 4 weeks and persisted for 12 weeks. In conclusion, shock wave therapy induces the ingrowth of neovascularization associated with early release of angiogenesis- related markers at the Achilles tendon-bone junction in rabbits. The neovascularization may play a role to improve blood supply and tissue regeneration at the tendon-bone junction.
Introduction
In clinical application, extracorporeal shock wave therapy has shown effect in the treatment of certain orthopedic conditions including non-union of long bone fracture [21,30,32,37,39], calcifying tendonitis of the shoulder [15,27,3 1,33,42], lateral epicondylitis of the elbow [ 10,13,24,28,34], proximal plantar fasciitis [6,19,20, 25,291 and Achilles tendonitis [24]. The success of shock wave therapy in our experiences ranged from 80'%1 for non-unions of long bone fractures [39] to 90'% for tendinopathies of the shoulder, elbow and heel [6,13,42]. In patients with calcifying tendonitis of the shoulder, radiographs showed that 58% of the calcium deposits were completely eliminated after shock wave therapy [42]. In addition, the short-term results of shock wave therapy for avascular necrosis of the femoral head appeared encouraging [16]. Shock wave therapy also showed a positive effect in promoting bone healing in animal experiments.
Despite the success in clinical application, the exact mechanism of shock wave therapy remains unknown. In fracture non-unions, it was speculated that shock wave therapy produces micro-fracture which in turn causes hematoma formation and subsequent callus formation and eventual fracture healing [7,11,14,22]. For tendino- pathy, it was postulated that shock wave therapy provokes painful level of stimulation, and relieves pain due to tendinopathy at the tendon-bone junction by hyper-stimulation analgesia [14,17,22,23,26,28,33,34, 371. However, there were insufficient data to support either theory. The results of our experiments in dogs demonstrated that shock wave therapy enhanced neo- vascularization at the tendon-bone junction [41]. Neo- vascularization was reported to be in association with painful tendinosis and may be responsible for pain in chronic Achilles tendinosis [3,25]. However, we specu- lated that the reasons for the dissolution of calcium deposits after shock wave therapy in calcifying tendo- nitis of the shoulder may involve a molecular mecha- nism of absorption due to improved blood supply. We further hypothesized that shock wave therapy may have the potential to induce the ingrowth of neovasculariza- tion and improvement of blood supply that lead to tis- sue regeneration. The purpose of this study was to investigate the effect of shock wave therapy on neovas- cularization at the tendon-bone junction of the Achilles tendons in rabbits.
Materials and methods
animals and shock wave treatment
This study was approved by the Institutional Review Board, and was performed under the guidelines and care of animals in research. Fifty New Zealand white rabbits with body weight ranging from 2.5 to 3.5 kg were used in this study. The right limb received shock wave therapy to the Achilles tendon near the insertion site to the heel bone, and was designated as the study side. The left limb received no shock wave therapy, and was designated as the control side. The rabbit was anesthetized with ketamine (50 mg/kg) and phe- nobarbital (30 mglkg) for the purpose of receiving shock wave appli- cation. The rabbit was placed in prone position with the right heel up. The shock wave tube was focused on the Achilles tendon near the insertion site to the heel bone, and the depth of the treatment was determined by the control guide and confirmed with C-arm image. Surgical lubricate was applied to the area of skin in contact with the shock wave tube. Each of the study limb received 500 impulses of shock waves at 14 kV (equivalent to 0.12 mJ1mm') in 20 min to the right Achilles tendon near the insertion site. The shock wave dosage so selected was based on the experience of our previous animal studies [40.41,44]. Immediately after shock wave therapy, the right limb was checked for swelling, edema, hemorrhage and motion of the limb. No special protection on the right limb or restriction of activities was provided. The rabbit was returned to the housing cage and was cared for by a veterinarian.
Histomorpholigical
assessment Biopsies of the Achilles tendon-bone units were performed in 0, I, 4. 8 and 12 weeks with 10 rabbits at each time interval. The first biopsy was obtained in 24 h after shock wave therapy. The specimens were fixed in 4'%1 buffered paraformaldehyde for 48 h and decalcified in PBS- buffered I 0'%, EDTA. Decalcified specimens were longitudinally cut into 5-pm sections and transferred to poly-lysine-coated slides. The number of blood vessels including capillaries and inusculariml vessels were examined microscopically, and quantitatively assessed in six areas of tendon-bone junction from three sections of the biopsy specimens. A professional pathologist blinded to the treatment regimen performed the measurements on all sections under 40x magnification
Immunohistochemistry
For the purpose of confirming neovascularization. the angiogenic markers including vessel endothelial growth factor (VEGF) and en- dothelial nitric oxide synthase (eNOS) were examined microscopically with imniunohistochemistry stains to reflect the mediators for neo- vascularization [5,35]. Detection of proliferating cell antigen (PCNA) was chosen to reflect endothelial cell replication [ 191. Immuno-reac- tivity in specimens was demonstrated using horseradish peroxidase (HRP)-3'-, 3'-diaminobenzidine (DAB) cell and tissue staining kil (R&D Systems. Inc. Minneapolis. MN, USA) in accordance with manufacturer's instructions. After blocking endogenous peroxidase and non-specific binding, sections were incubated overnight with the antibodies at 4 "C. Polyclonal antibodies used for immunohisto- chemistry were anti-eNOS, anti-VEGF and anti-PCNA (Upstate Biotechnology. Lake Placid, NY, USA). Sections were further incu- bated with biotinylated anti-goat IgG and then incubated with strep- tavidin conjugated to HRP. Immuno-reactivity was determined by incubating the sections in a chromogen solution containing DAB and 0.1% hydrogen peroxide in the dark, followed by counterstaining with hematoxylin. Dehydrate sections were mounted with mounting me- dium. Those without primary antibodies were enrolled as negative controls for the immunostaining. Vessels showing positive VEGF ex- pression and cells displaying positive PCNA and eNOS expressions were counted microscopically. The numbers with positive expression were quantitatively assessed, and the averages were calculated from six areas of tendon-bone junction selected from three sections of each biopsy specimen.
statistrical analysis
Data were presented as mean k standard error. The numbers of vessels and cells dispbaying positive angiogenic markers at each time interval between the study side and the control side were compared statistically using Mann-Whitney test with statistical significance at p < 0.05.
Results
Increase of' neo-vessels after shock wave treutment
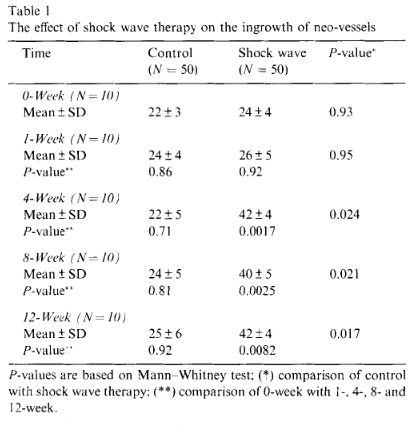
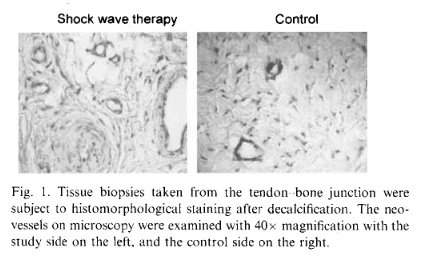
The results of neo-vessels of the study and the control sides are summarized in Table 1. In the study side, a significant increase of neo-vessels was noted in 4 weeks, and the increase in neo-vessels persisted until 12 weeks, whereas no increase of neo-vessels was noted in the control side, and the difference in the number of neo- vessels was statistically significant. The microscopic features of neo-vessels of the study and the control sides are shown in Fig. I. It appeared that formation of neo- vessels after shock wave therapy was time dependent starting in 4 weeks after treatment and it lasted for 12 weeks or longer.
Increase of ungiogenic markers ujter shock wave therapy
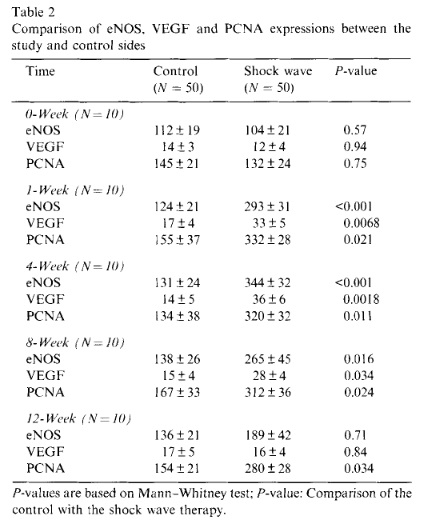
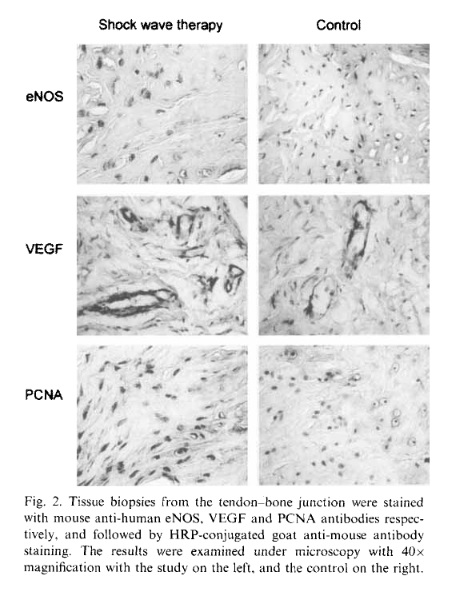
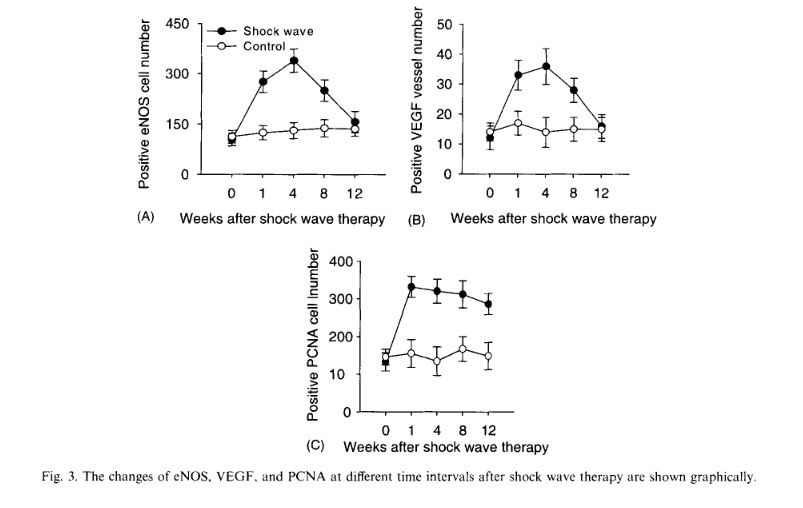
The results of angiogenesis-related markers including eNOS, VEGF and PCNA are summarized in Table 2. In the study side, a significant increase in eNOS, VEGF and PCNA was noted in as early as one week. The in- crease of eNOS and VEGF lasted for 8 weeks before they declined to normal at 12 weeks, whereas the in- crease of PCNA lasted until 12 weeks. In the control side, however, no significant changes were noted in eNOS, VEGF and PCNA, and the differences between the study and control sides are statistically significant. The microscopic features of eNOS, VEGF and PCNA expressions of the study and the control sides are shown in Fig. 2. The changes in eNOS, VEGF and PCNA expressions at different time intervals are graphically illustrated in Fig. 3. The results of this study demon- strated that shock wave therapy induces early expres- sions of eNOS, VEGF and PCNA in one week, and subsequent ingrowth of neo-vessels in approximately 4 weeks. The increases of eNOS and VEGF were transient and only lasted for 8 weeks; whereas the stimulation of cell proliferation and the ingrowth of new vessels per- sisted for 12 weeks and longer. The biological responses after shock wave therapy appear to be time-dependent.
Discussion
Many authors had studied the mechanism of shock wave therapy [7-9,11,12,14,17,18,22-24,26,28,33,34,36, 38,40,41,4346]. Some speculated that shock wave therapy relieves pain due to insertional tendinopathy by hyper-stimulation analgesia [14,22-24,28,37], while others hypothesized the theoretical mechanism of shock wave therapy in bone healing including micro-disrup- tion of avascular or minimally vascular tissue to encourage revascularization and the recruitment of ap- propriate stem cells conductive to more normal bone tissue healing [ 1 I, 12,14,18,22,4346]. However, there are insufficient data to scientifically substantiate either theory concerning the mechanism of shock wave therapy in musculoskeletal disorders. The etiologies of tendonitis are multi-factorial in- cluding degenerative changes, inflammatory process and metabolic disturbance with hypo-vascularity, neu- ronal origin as well as neovascularization in tendinosis [14,24,25]. Neovascularization was found to be in close relation to the area of tendinosis that corre- sponded to the painful area during Achilles loading. Therefore, neovascularization, rather than a “healing response”, was thought to be responsible for the pain in the area with chronic Achilles tendinosis [3,25]. On the contrary, neovascularization, a direct effect of angio- genic factors secondary to extracorporeal shock wave therapy was found to be responsible for improvement in symptoms of plantar fasciitis [33,34]. In calcifying tendonitis of the shoulder, shock wave therapy not only provides pain relief, it also eliminates calcium deposits [15,26,27,42]. The mechanical forces of shock wave therapy may theoretically cause fragmentation, but not total absorption of the calcium deposits. Therefore, it is reasonable to assume that a biological process through a molecular mechanism of absorption may take place to dissolve the calcium deposits after shock wave therapy. Nitric oxide and VEGF had been demonstrated as important mediators of angiogenesis [5,35]. We suggest that physical shock waves could raise the mechanotransduction of the tissue to convert acoustic shock wave energy into biological signals. The results of this study showed that shock wave therapy induces the ingrowth of neo-vessels and tissue prolif- eration associated with the early release of angiogene- sis-related factors including eNOS and VEGF at the tendon-bone junction in rabbits. Therefore, the mech- anism of shock wave therapy appears to involve the early release of angiogenic growth factors in one week, and induces cell proliferations and formation of neo- vessels in approximately four weeks at the tendon- bone junction. The neovascularization may lead to the improvement of blood supply and play a role in tissue regeneration at the tendon-bone junction. No adverse effect of neovascularization on rabbits was observed during the course of this study. In non-unions of long bone fractures, the fracture sites are generally filled with dense, hypo-vascular fibrous tissues that prevent the fractures from bony union. Many studies in animal experiments had dem- onstrated that shock wave therapy can promote bone marrow stromal cell differentiation toward osteopro- genitors associated with induction of TGF-P1 and in- duces membrane hyperpolarization and Ras activation to act as an early signal for the osteogenesis in human bone marrow stromal cells [36,4346]. Other studies also showed a positive effect of shock wave therapy in promoting bone healing in animal experiments [8,11,12,18]. The results of this study showed that shock wave therapy promotes early release of angio- genic factors, and subsequently induces cell prolifera- tions and ingrowth of neo-vessels that in turn may stimulate the stromal cell growth and differentiation and promote bone healing. In conclusion, shock wave therapy induces the in- growth of neovascularization and cell proliferation as- sociated with earlier release of angiogenic growth factors at the tendon-bone junction of the Achilles tendon in rabbits. It appears that the mechanism of shock wave therapy involves the early release of angiogenic growth factors (eNOS and VEGF) aiid subsequent induction of neovascularization and tissue proliferation. The neo- vascularization may play a role in pain relief of tendi- nitis and the repair of chronically inflamed tendon tissues at the tendon-bone junction.
References
- Ackerman PW, Jim L, Finn A, Ahmed M, Kreicbergs A. Autonomic innervation of tendons, ligaments and joint capsules: A morphologic and quantitative study in the rat. J Orthop Res 2001:19:372-8.
- Ackermaii PW. Ahnied M, Kreicbergs A. Early nerve regenera- tion after Achilles tendon rupture-a prerequisite for healing. A study in the rat. J Orthop Res 2002:20:849-56.
- Alfredson H, Bjur D, Thorsen K, Lorentzon R. High intratendi- nous lactate levels in painful tendinosis. An investigation using microdialysis technique. J Orthop Res 2002:20:934-8.
- Archer RS, Bayley JI. Acher CW, et al. Cell and matrix changes associated with pathological calcification of the human rotator cuff tendons. J Anat 1993:182:1-11.
Babaei S. Stewart DJ. Overexpression of endothelial NO synthase induces angiogenesis in a co-culture model. Cardiovascular Res 2002;55: 190-200. - Chen HS. Chen LM. Huang TW. Treatment of painful heel syndrome with shock waves. Clin Orthop 2001:387:41-6.
 Tecar
Tecar Shock Wave
Shock Wave Plasma
Plasma Laser
Laser Tesla
Tesla Nikola
Nikola Kayser
Kayser Fusion
Fusion