Extracorporeal shock wave therapy in early osteonecrosis of the femoral head: prospective clinical study with long-term follow-up
- Type: Free
Introduction Extracorporeal shock wave therapy (ESWT) may exert beneWcial eVects in avascular necrosis of femoral head (AVNFH). Patients The current study evaluated the eVectiveness of ESWT in reducing pain and in slowing down the progression of bone damage in 36 patients with unilateral AVNFH of stage Association Research Circulation Osseous (ARCO) I, II and III. At the beginning of the study, 10 hips were classiWed as stage I, 11 as stage II and 15 as stage III. Each treatment cycle included four sessions, with 2,400 impulses each administered at 0.50 mJ/mm2, at 48–72 h intervals. Follow-up examinations were scheduled at 3, 6, 12 and then 24 months. Method Clinical assessments included assessment of pain scores, Harris Hip Scores and Roles and Maudsley score. Plain radiographs and magnetic resonances of the hip were used to evaluate the size of the lesion, the extent of collapse of subchondral bone, and degenerative changes of the hip joint. Results Patients from ARCO stage I group and stage II group achieved signiWcantly better results than patients from ARCO stage III group at all follow-up time points (p < 0.005). During the follow-up period, 10 of the 15 stage III ARCO patients received an arthroplasty. ARCO stages I and II lesions were unchanged on radiographs and on magnetic resonance images. Conclusion ESWT in ARCO stages I and II may help to prevent progression of the area of avascular necrosis and manage pain.
Keywords Shock wave therapy · Avascular necrosis of femoral head · ESWT · AVNFH · Conservative treatment
Introduction
Avascular necrosis of femoral head (AVNFH) is caused by insuYcient blood supply, and is histologically associated with death of osteocytes, followed by osteoclastic resorption of dead trabeculae and apposition of new bone tissue [1]. AVNFH aVects more than 10,000 individuals in the USA each year, and is more common in males than females with a ratio 7–3, with the exception of cases associated with
systemic lupus erythematosus [2, 3]. In a current model of AVNFH, an ischemic attack is followed by increased intraosseous pressure, probably from the edema in the bone marrow compartment, which is functionally closed, so that the venules and capillaries are constantly compressed. This leads to the establishment of a vicious circle similar to compartment syndrome of a limb.
Avascular necrosis of femoral head is assessed using plain anterior–posterior and lateral or “frog leg” radiographs. While radiographs have high speciWcity for advanced disease but a low sensitivity for early AVNFH, 99Tc bone scintigraphy is highly sensitive in the early stages of the disease, with the characteristic “cold to hot” appearance [4]. MRI remains the most important diagnostic aid, with a sensitivity of 99% and a speciWcity of 98% [5]. In T1-weighted sequences, there is a low signal line between healthy bone and ischemic bone, known as “bandlike lesion”, which corresponds to a zone of sclerosis and
Wbrosis. T2-weighted sequences show evidence of a second inner line of high signal (double line sign), indicative of hypervascularity resulting from the repair process [4]. The international classiWcation system of the Association Research Circulation Osseous (ARCO) [6] was introduced in 1993, and has been proposed as a new classiWcation system which includes the previous classiWcations [7].
Management options for AVNFH remain controversial. For the early stages of AVNFH, femoral head preserving procedures are recommended, including core decompression, vascularized or non- vascularized bone graft and osteotomy, while in advanced stages (ARCO III and IV) total hip arthroplasty is usually performed [8]. Conservative management modalities used in AVNFH include prostacycline- analogue iloprost, enoxaparin treatment and alendronate [9–12]. The most used physical therapies are pulsed electromagnetic Welds (PEMFs), which seem to be able to control inXammatory processes and facilitate the reparative rocesses of the bone, and extracorporeal shock wave therapy (ESWT), which can activate many cellular processes critical to neovascularization and tissue regeneration [8, 13–20]. This longitudinal study evaluated the eVectiveness of high energy ESWT in reducing pain and in slowing down the progression of bone damage in patients with AVNFH of stage ARCO I, II and III.
Materials and methods
Between April 2003 and March 2009, we conducted this prospective, observational clinical study. The study protocol was approved by our Institutional Review Board. Recruitment of patients and data monitoring were performed in our university hospital (Sapienza University School of Medicine), by a physician not involved in the selection and treatment of the patients enrolled in the present investigation. Inclusion criteria were stage I, II, or III AVNFH according to the ARCO international classiWcation system. Exclusion criteria included skeletal immaturity, a IV ARCO stage lesion, current or previous infection, cardiovascular and neoplastic disease, an implanted pacemaker, blood coagulation disorders, use of anticoagulant drugs and pregnancy. All patients gave written informed consent to participate in the study. Thirty-six patients (23 males and 13 females) met the inclusion criteria and were recruited for this study (Table 1).
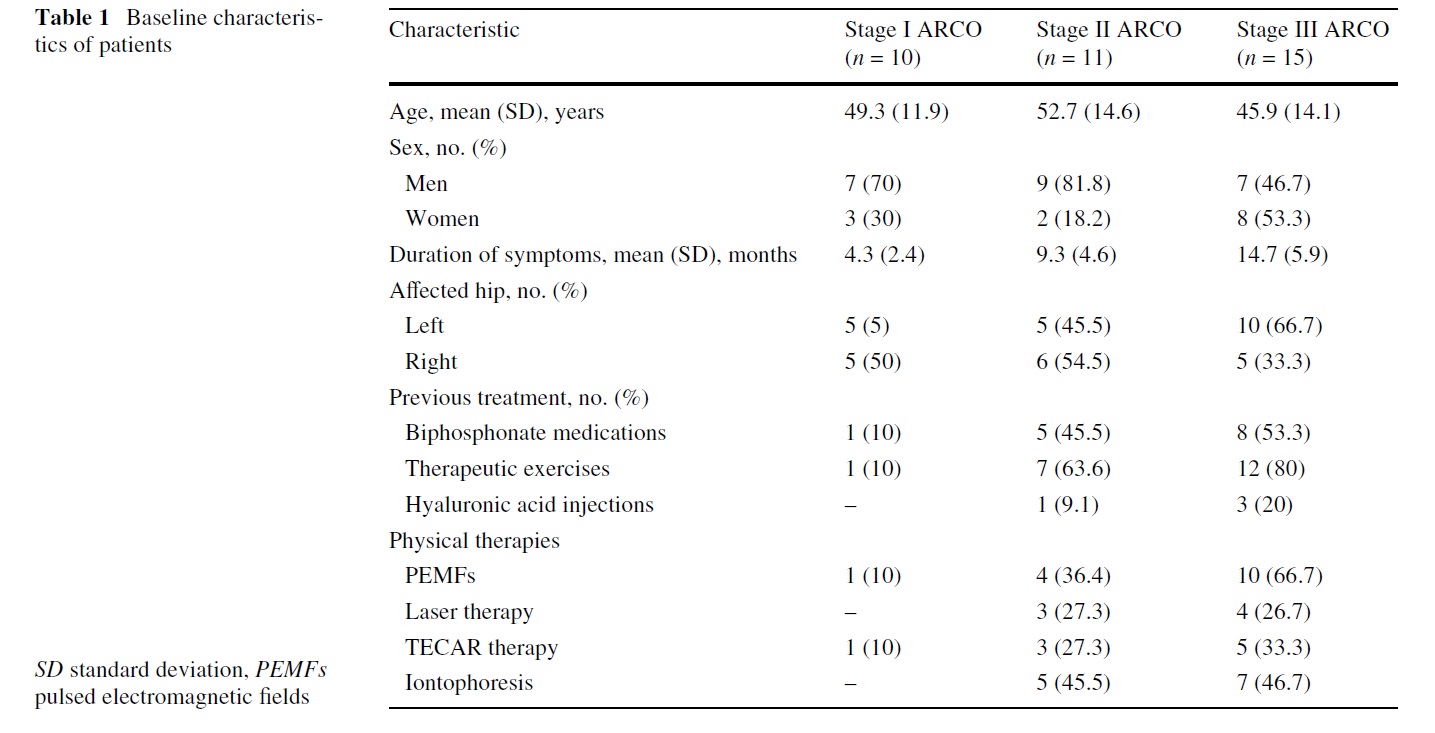
The diagnosis of AVNFH was conWrmed with plain radiographs and magnetic resonance imaging (MRI). There were 18 patients without risk factors and therefore classi- Wed as idiopathic osteonecrosis, 2 patients with secondary congenital hip dysplasia, 4 patients with a history of acute traumatic events (2 with hip dislocation). In the remaining patients, identiWed risk factors were smoking and previous
corticosteroid therapy. Patients were staged according to ARCO classiWcation: at the beginning of the study 10 hips were classiWed as stage I, 11 as stage II and 15 as stage III. Pre-treatment assessments consisted of a complete history and physical examination, MRI and radiographs of the aVected hips. Other factors taken into account were objective activity restriction, mobility and ability to perform the activities of daily living.
Shock wave treatment
The shock weave treatment was applied using an electromagnetic shock wave generator (Modulith SLK, STORZ MEDICAL AG, Switzerland), with a penetration depth between 0 and 150 mm and a focus diameter of 4 mm. Shock waves were focused around (on the margins of) the necrotic bone of the femoral head under radiographic guidance. The treatment area was prepared with a coupling gel to minimize the loss of shock-wave energy at the interface between the head of the device and the skin. All ESWT procedures were performed without general or regional anaesthesia. The administering physician was experienced in the use of extracorporeal shock-wave therapy in the treatment of various musculoskeletal disorders. Each treatment cycle included four sessions, with 2,400 impulses each administered at 0.50 mJ/mm2, at 48–72 h intervals. After ESWT treatment, patients were instructed to walk on crutches and not to bear weight on the aVected limb for 2 months. No other therapies were administered to any patient. Patients who did not respond to ESTW and those dissatisWed with the treatment results underwent total hip arthroplasty.
Outcome measures
Follow-up examinations were scheduled at 3, 6, 12 and then 24 months. Clinical assessments included assessment of pain scores, Harris Hip Scores and Roles and Maudsley score. The intensity of pain was recorded on a visual analogue scale (VAS), ranging from 0 to 10, with 0 indicating no pain and 10 indicating severe pain. The Harris Hip Score gives 91 points for assessment of pain and function and 9 points for range of motion and deformity [21]. The Roles and Maudsley score is a subjective 4-point patient assessment of pain and limitations of activity, and has been used extensively to assess outcome after ESWT [22]. On this scale, 1 point is deWned as an “excellent” result with the patient having no symptoms. Two points is deWned as a “good” result, with the patient signiWcantly improved from the pre-treatment condition and satisWed with results. Three points is deWned as a “fair” result with the patient somewhat improved from pre-treatment condition and partially satisWed with treatment outcome. Four points indicates a “poor” outcome with symptoms identical or worse than pre-treatment condition and with dissatisfaction with treatment results. In the present study, a hip converted to a total hip arthroplasty at any time during the study was also deWned as “poor” outcome. Antero-posterior and lateral radiographs were made before treatment, and at 3, 6, 12 and 24 months after treatment. Plain radiographs of the hip were used to evaluate the size of the lesion, the extent of collapse of subchondral bone and degenerative changes of the hip joint. MRI was performed before treatment, at 6 and 12 months after the end of treatment, and once a year thereafter, using axial, coronal, sagittal T1 and T2-weighted scans, STIR and T2 with fat saturation sequences. These images were used to measure the size of the lesion, assess the congruency of the femoral head, the presence of a crescent sign and/or degenerative changes of the hip joint, with the aim to stage patients’ hips according to ARCO classiWcation. Radiographic assessments and clinical examinations were performed, respectively, by two independent radiologists and two clinicians not involved in the treatment procedures. Every discrepant evaluation was discussed, and a decision was reached by consensus.
Statistical analysis
The aim of this study was to evaluate the clinical outcome and the diagnostic changes in each ARCO stage group and between the groups after focused ESWT at Wve time points (before treatment and at 3, 6, 12 and 24 months). The eYcacy endpoints were prospectively deWned as: reduction of the self-rated pain intensity scale (VAS), improvement of the Harris Hip Score, reduction of the Roles and audsley score and regression or stabilization of ARCO stage. Descriptive statistics were applied for all ARCO stage groups. Continuous variables were summarized within ARCO groups using mean and standard deviation. The paired samples t-test was used for comparison of mean change of the VAS and the Harris Hip score between the pre-treatment and scheduled follow-up time points within
each ARCO group. One-way analysis of variance was used to assess whether there were signiWcant diVerences in the VAS and Harris Hip Score among the three groups at various follow-up periods. The Wilcoxon rank-sum test and the Mann–Whitney U-test were used respectively for comparing changes of the Roles and Maudsley score within each group between pre-treatment and post-treatment assessments, and between groups at each evaluation time. Data were analyzed using SPSS for Windows (version 16.0; SPSS, Chicago, III). p Values less than 0.05 were considered statistically signiWcant.
Results
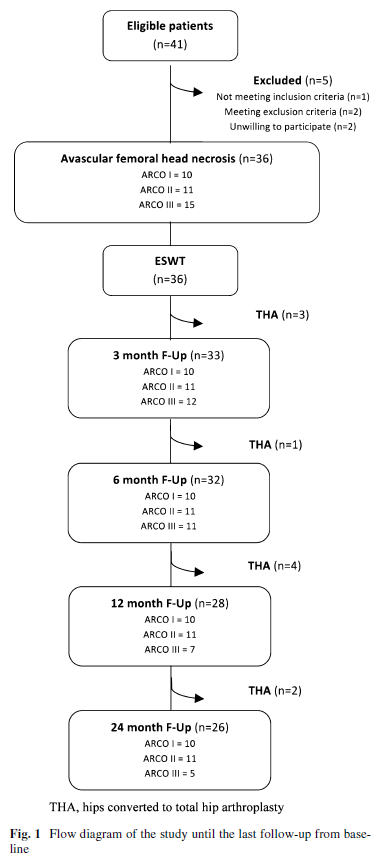
Of the 36 hips which underwent shock wave treatment, 3 failed and were converted to total hip arthroplasty within the 3-month follow-up because of persistence of pain at weight-bearing and functional limitations. In the following months, in another seven patients a total hip arthroplasty was necessary: one before the 12 month follow-up, four before the 24 month follow-up and two after 24 months.
All the subjects who received an arthroplasty were at ARCO III stage. They were not followed up after conversion to a total hip arthroplasty. We, therefore, evaluated 33 hips at the Wrst follow-up (3 months), 32 hips at the second follow-up (6 months), 28 hips at the third follow-up (12 months) and 26 hips at the last evaluation (24 months).
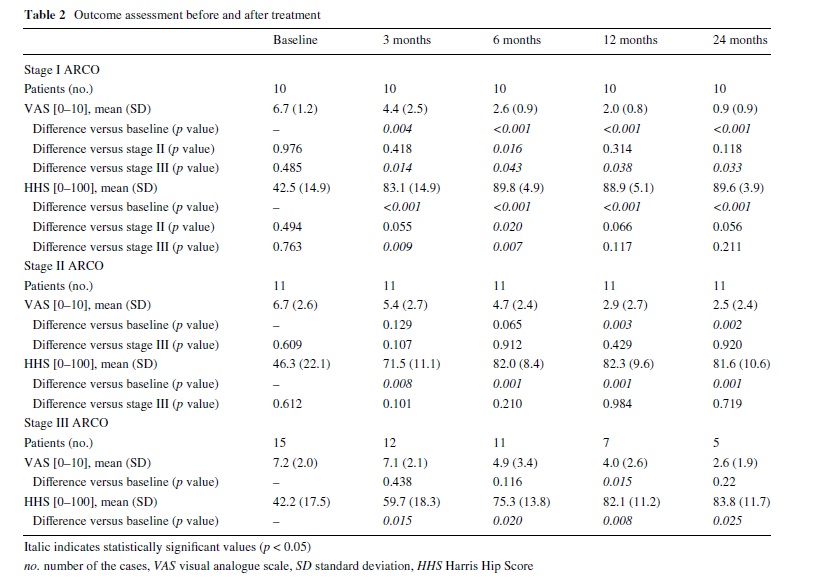
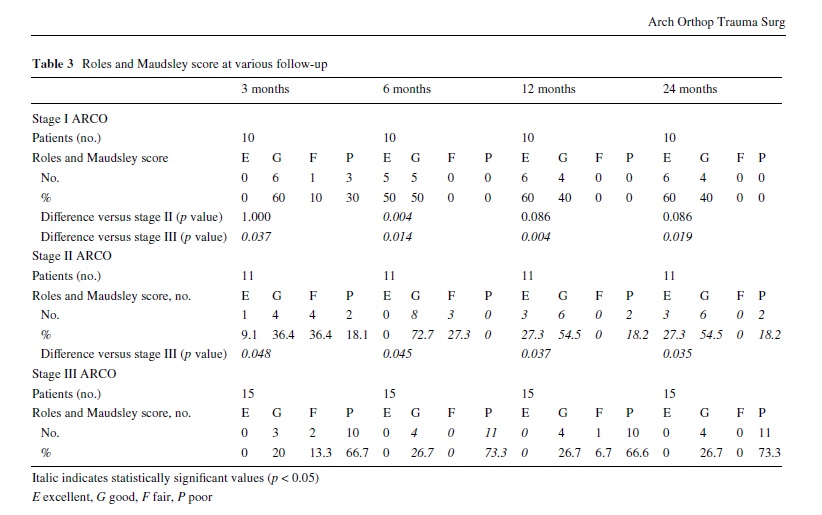
The evolution of the population at all follow-up time points was summarized in Fig. 1. Outcome measures (scores on the VAS, Harris Hip Score and Roles and Maudsley score) are shown in Tables 2 and 3. At all follow-up periods, stage I ARCO patients showed signiWcantly better results in VAS score (all p < 0.001) than before treatment. In ARCO stages II and III groups, signiWcant reduction of the VAS was noted only at third and fourth follow-up time points after ESWT treatment (Table 2). Conversely, the Harris Hip Score showed statistically signiWcant improvement at various follow-up time points from pre-treatment values for all treatment groups (all p < 0.001): ARCO stage I patients obtained satisfactory values (mean score > 75) from the Wrst follow-up, while in the other two groups this mean score was obtained only from the second follow-up (6 months after treatment). The VAS score in ARCO stage I group was signiWcantly lower than in ARCO stage II group at 6 months (p = 0.016), whereas it was signiWcantly better in ARCO stage I group than in ARCO stage III group at all follow-up time points (Table 2).
Association Research Circulation Osseous stage I patients showed signiWcantly better results in Harris Hip Score than ARCO stage II group at second follow-up period (p = 0.02), and than ARCO stage III group at 3 and 6 months (respectively, p = 0.009 and p = 0.007). No signiWcant diVerences in the mean VAS and Harris Hip Score were observed between ARCO stages II and stage III groups at any follow-up period (all p > 0.05).
At pre-treatment evaluation, all patients rated the condition of the aVected hip as “4” (poor) in the subjective four point Roles and Maudsley score. The 10 ARCO stage III patients submitted to total hip arthroplasty at various follow- up were deWned as “4” (poor) outcome. No patient reported worsening of symptoms compared to pre-treatment (Table 3). The diVerence between the ARCO stage I and stage II patients was signiWcant at 6 months (p = 0.004). The diVerence in the success rate between the two groups was no longer signiWcant. Patients from ARCO stage I group and stage II group achieved signiWcantly better results than patients from ARCO stage III group at all follow-up time points. Overall, at 3 months follow-up, 6 of 10 patients (60%) in ARCO stage I group, 5 of 11 patients
(45.5%) in stage II group and 3 of 15 patients (20%) in stage III group reported a 1 (excellent result) or 2 (good result) score on the Roles and Maudsley score; at 6 months follow-up, all the 10 patients (100%) in ARCO stage I group, 8 of 11 patients (72.7%) in stage II group and 4 of 15 patients (26.7%) in stage III group reported excellent or good results; at 12 and 24 months follow-up, all the 10
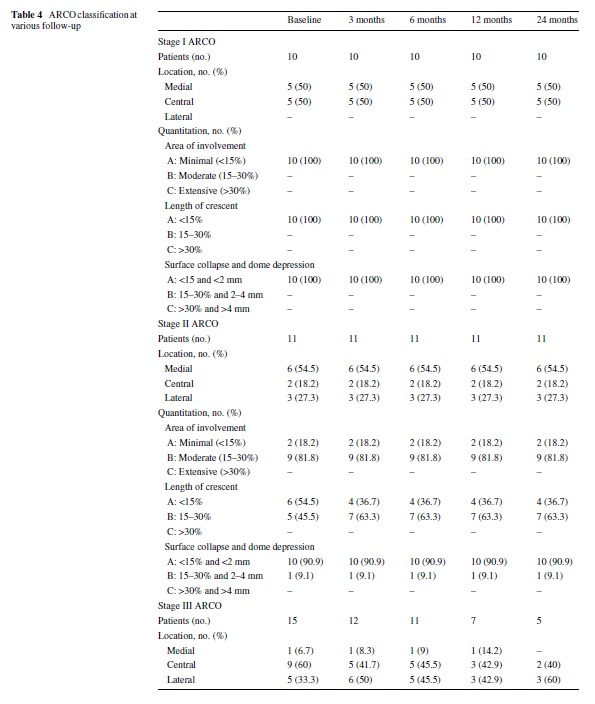
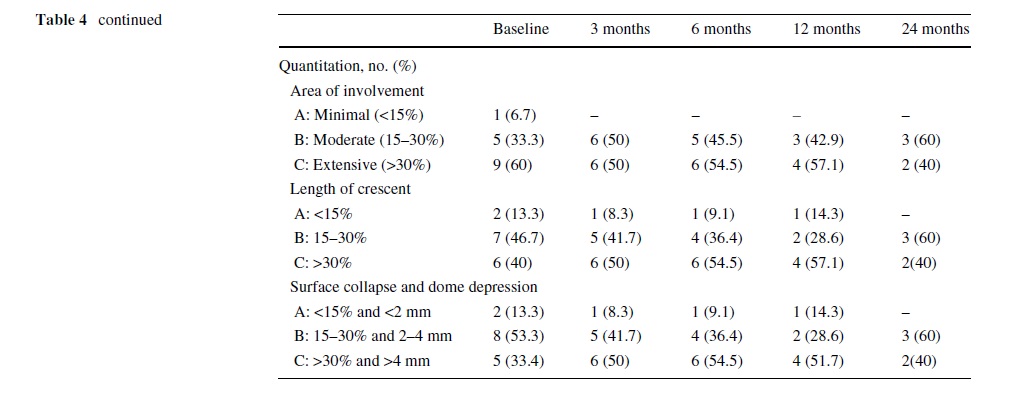
patients (100%) in stage I group, 9 of 11 patients (81.8%) in stage II group and 4 of 15 patients (26.7%) in stage III group reported excellent or good results. At all follow-up time points, the lesions show no or only minimal changes on radiographs and on magnetic resonance images. Neither regression nor progression of lesions that had been graded before treatment as ARCO stage I and II were seen despite clinical improvement. However, in 10 of 15 ARCO stage III patients, there was clinical deterioration and the patients had a THA. In the rest of Wve patients, minimal radiographic or MRI changes were observed (Table 4).
Side effects
clinically detectable neuromuscular, systemic or devicerelated adverse eVects were observed. In all patients, only minor complications occurred after ESWT, such as transient soft tissue swelling or minor bruising. No other adverse eVects were noted.
Discussion
Hips with AVNFH usually deteriorate, with collapse of the femoral head, leading ultimately to joint destruction and the need for arthroplasty. Ohzono et al. [23] reported the collapse of the femoral head in 94–100% of cases if the lesions are at the weight-bearing surface on the femoral head within 5 years.Management of AVNFH remains controversial, and, if
conservative measures fail, surgery is indicated. Non-surgical approaches to early AVNFH include protected weightbearing and physical therapies, such as PEMFs or focused ESWT. PEMFs may limit local inXammation, preventing degeneration of the articular cartilage since they reduce the production of oxygen free radicals, limiting the catabolic eVects of inXammatory cytokines on the articular cartilage
by promoting the synthesis of proteoglycans, stimulate the reparative processes and may enhance the healing process by promoting neovascularization and the formation of new bone. These combined eVects can be eVective in pain control, for chondroprotection, and for optimization of the healing process [13, 14].
The exact mechanism through which ESWT operates remains unknown. In mice, ESWT regulate the expression of endothelial growth factor (VEGF) and BMP-2. The increase in the expression of VEGF and BMP-2 was evaluated by two diVerent trials conducted in 2007 and 2008, respectively, using the same technique. These studies were performed on mice in which osteonecrosis had been induced after bilateral administration of methylprednisolone and lipopolysaccharide, and in which treatment with ESWT was performed 6 weeks after corticosteroid, at 2,000 pulses with a level of energy of 0.26 mJ/mm2. Necrosis was induced bilaterally to use the left limb for treatment with shock waves and the right limb as control. Ma et al. [15] postulated that VEGF might be involved in the positive eVects of shock wave therapy
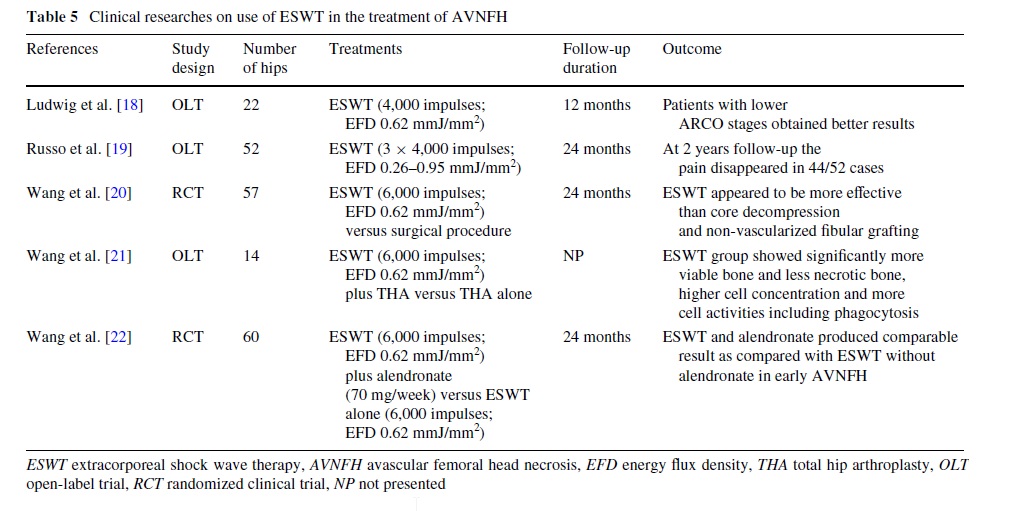
because VEGF is a speciWc mitogenic factor for vascular endothelial cells, which stimulates endothelial cells proliferation, promotes neovascularization and increases vascular permeability15. The increased expression of BMP-2 in femoral heads treated with ESWT was a key Wnding because the BMP-2 is a key mediator of bone development and repair for its capacity to mobilize osteoprogenitor cells, thereby promoting osteoblastic diVerentiation processes and resulting in bone formation [16]. Other eVects of ESWT are: increase of the endothelial nitric oxide synthase, thereby promoting neovascularization, the increase of endorphins resulting in pain control and increase in other growth factors such as EGF, IGF 1 and PDGF. Finally, Wang et al. [24] demonstrated that local mechanical stimulation with shockwave may reXect the systemic eVects of angiogenesis and osteogenesis and anti-inXammation in hips with AVNFH24. Recently, Hausdorf et al. [25] also showed that ESWT are able to propagate through the femoral head with a distance-related absorption and a pressure loss of about 50% within 10 mm of bone, and they stated that the shock wave energy used in clinical studies seems to be suYcient to produce biological responses in terms of vascular and bone growth. The beneWcial eVects of ESWT in AVNFH has been conWrmed by several recent clinical studies (Table 5).
ESWT is eVective in pain reduction, and in improving hip function and, consequently, the quality of life of patients with AVNFH, with better results in the early stages of the condition (ARCO stages I and II) [8, 18–20]. In the literature, some studies compared conservative and surgical management in AVNFH. One investigation showed that ESWT appeared to be more eVective than core decompression
and non-vascularized Wbular grafting [19]. Finally, ESWT and alendronate may have synergistic eVects through diVerent mechanisms in early AVNFH [20].
Our results conWrmed these Wndings. In fact, 10 of 15 ARCO stage III patients underwent total hip arthroplasty during the follow-up period because of persistence of hip pain and functional limitations. These subjects were considered as poor outcome on Roles and Maudsley score where, already at 6 months follow-up time, all patients in ARCO stage I group and 8 of 11 patients (72.7%) of subjects in stage II group reported excellent or good results. Conversely, during follow-up assessments, only 4 of 15 patients in stage III obtained good results and none of them reported excellent outcomes.
Furthermore, we observed a signiWcant reduction of mean VAS score at short-, medium-- and long-term followup only in ARCO stage I patients, whereas the mean Harris Hip score showed statistically signiWcant improvement at all follow-up time points from pre-treatment values for all treatment groups. Thus, the osteonecrosis progression appears to be the main factor in determining pain, but pain
is not the only cause that aVects hip functions in people with AVNFH. Finally, during the follow up period all hips with early AVNFH (ARCO I and II) treated with ESWT alone appeared only minimal radiographic and MRI changes and overall a clinical improvement. Therefore, as the natural history of AVNFH is of progression, the beneWcial eVects of ESWT for the early stages, (ARCO I and II),
is evident in the fact that the AVNFH area involved did not increase and the grade of it did not worsen. Furthermore, the patients had clinical improvement which was mostly obvious after the Wrst 6 months of ESWT treatment.
We fully acknowledge the limitations in this study. For example, we have reported on a relatively small series of patients, and we lack a control group. However, in our setting, AVNFH is relatively uncommon, and it would have been diYcult to mount a randomized controlled trial. Another limitation is that some patients were already treated on diVerent methods previously and not with a very
big gap before their ESWT beginning, as pointed out from Table 1. Finally, the comparison of results obtained in our study with other studies using ESWT is only partially possible, given the diVerences in the type of equipment used, treatment and evaluation protocols, inclusion criteria and number of patients treated. Given the considerable impact of AVNFH on public health, multicenter studies should be conducted on the eVectiveness of ESWT in the management of avascular necrosis, to collect larger numbers of patients treated in a homogeneous fashion and assessed with the same methodology.
Conclusion
High energy ESWT can be eVective in the management of AVNFH ARCO stages I and II, but less so in the more advances stages of the condition. Early diagnosis and staging are, therefore, of extreme importance to obtain beneWts from ESWT. Additionally, ESWT is non-invasive, has a low complication rate, does not require hospitalization and has a relatively low cost compared to other types of conservative and surgical approaches, with a relatively short time of application. Our results also conWrm reports in the literature indicating that ESWT in ARCO stages I and II aims to slow down the worsening of the grade of the AVNFH in the avascular area and improve clinical features. Further studies are needed with larger cohorts of patients using the homogenous classiWcation system, and standardized treatment protocols to further assess the eVectiveness of ESWT in the management of AVNFH. The research was carried out in accordance with the ethical standards described by the Helsinki Declaration, and was approved by a local Ethical Committee.
References
1. Chandler F (1948) Coronary disease of the hip. J Int Coll Surg 11:34–36
2. Mankin HJ (1992) Nontraumatic necrosis of bone (osteonecrosis). N Engl J Med 326(22):1473–1479
3. Assouline-Dayan Y, Chang C, Greenspan A, Shoenfeld Y, Gershwin ME (2002) Pathogenesis and natural history of osteonecrosis. Semin Arthritis Rheum 32(2):94–124
4. Malizos KN, Karantanas AH, Varitimidis SE, Dailiana ZH, Bargiotas K, Maris T (2007) Osteonecrosis of the femoral head: etiology, imaging and treatment. Eur J Radiol 63(1):16–28
5. Theodorou DJ, Malizos KN, Beris AE, Theodorou SJ, Soucacos PN (2001) Multimodal imaging quantitation of the lesion size in osteonecrosis of the femoral head. Clin Orthop Relat Res 386:54–63
6. Gardeniers JWM (1993) The ARCO perspective for reaching one uniform staging system of osteonecrosis. In: Scoutens A, Arlet J, Gardeniers JWM, Hughes SPF (eds) Bone circulation and vascularization in normal and pathologic conditions. Plenum, New York, pp 375–380
7. Steinberg ME, Hayken GD, Steinberg DR (1995) A quantitative system for staging avascular necrosis. J Bone Joint Surg Br 77(1):34–41
8. Wang CJ, Wang FS, Ko JY, Huang HY, Chen CJ, Sun YC, Yang YJ (2008) Extracorporeal shockwave therapy shows regeneration in hip necrosis. Rheumatology (Oxford) 47(4):542–546
9. Disch AC, Matziolis G, Perka C (2005) The management of necrosis- associated and idiopathic bone marrow edema of the proximal femur by intravenous iloprost. J Bone Joint Surg Br 87(4):560–564
10. Glueck CJ, Freiberg RA, Sieve L, Wang P (2005) Enoxaparin prevents progression of stages I and II osteonecrosis of the hip. Clin Orthop Relat Res 435:164–170
11. Lai KA, Shen WJ, Yang CY, Shao CJ, Hsu JT, Lin RM (2005) The use of alendronate to prevent early collapse of the femoral head in patients with nontraumatic osteonecrosis. A randomized clinical study. J Bone Joint Surg Am 87(10):2155–2159
12. Nishii T, Sugano N, Miki H, Hashimoto J, Yoshikawa H (2006) Does alendronate prevent collapse in osteonecrosis of the femoral head? Clin Orthop Relat Res 443:273–279
13. Massari L, Fini M, Cadossi R, Setti S, Traina GC (2006) Biophysical stimulation with pulsed electromagnetic Welds in osteonecrosis of the femoral head. J Bone Joint Surg Am 88(Suppl 3):56–60
14. Hopper RA, VerHalen JP, Tepper O, Mehrara BJ, Detch R, Chang EI, Baharestani S, Simon BJ, Gurtner GC (2009) Osteoblasts stimulated with pulsed electromagnetic Welds increase HUVEC proliferation via a VEGF—a independent mechanism. Bioelectromagnetics 30(3):189–197
15. Ma HZ, Zeng BF, Li XL (2007) Upregulation of VEGF in subchondral bone of necrotic femoral heads in rabbits with use of extracorporeal shock waves. Calcif Tissue Int 81(2):124–131
16. Ma HZ, Zeng BF, Li XL, Chai YM (2008) Temporal and spatial expression of BMP-2 in sub-chondral bone of necrotic femoral heads in rabbits by use of extracorporeal shock waves. Acta Orthop
79(1):98–105
17. Ludwig J, Lauber S, Lauber HJ, Dreisilker U, Raedel R, Hotzinger H (2001) High-energy shock wave treatment of femoral head necrosis in adults. Clin Orthop Relat Res 387:119–126
18. Russo S, Corrado EM, Corrado B et al (2004) The role of extracorporeal shock waves. In: Santori FS, Santori N, Piccinato A (eds) vascular necrosis of the femoral head: current trends. Springer,
Milan
19. Wang CJ, Wang FS, Huang CC, Yang KD, Weng LH, Huang HY (2005) Treatment for osteonecrosis of the femoral head: comparison of extracorporeal shock waves with core decompression and
bone-grafting. J Bone Joint Surg Am 87:2380–2387
20. Wang CJ, Wang FS, Yang KD, Huang CC, Lee MS, Chan YS, Wang JW, Ko JY (2008) Treatment of osteonecrosis of the hip: comparison of extracorporeal shockwave with shockwave and alendronate. Arch Orthop Trauma Surg 128(9):901–908
21. Harris WH (1969) Traumatic arthritis of the hip after dislocation and acetabular fractures: treatment by mold arthroplasty: an endresult study using a new method of result evaluation. J Bone Joint
Surg Am 51:737–755
22. Roles NC, Maudsley RH (1972) Radial tunnel syndrome: resistant tennis elbow as a nerve entrapment. J Bone Joint Surg Br 54:499–508
23. Ohzono K, Saito M, Sugano N, Takaoka K, Ono K (1992) The fate of nontraumatic avascular necrosis of the femoral head: a radiologic classiWcation to formulate progress. Clin Orthop 277:73–78
24. Wang CJ, Yang YJ, Huang CC (2010) The eVects of shockwave on systemic concentrations of nitric oxide level, angiogenesis and osteogenesis factors in hip necrosis. doi:10.1007/s00296-010-1384-7
25. Hausdorf J, Lutz A, Mayer-Wagner S, Birkenmaier C, Jansson V, Maier M (2010) Shock wave therapy for femoral head necrosis—pressure measurements inside the femoral head. J Biomech
doi:10.1016/j.jbiomech.2010.04.009
Related articles
Shock-wave therapy is effective for chronic calcifying tendinitis of the shoulder
 Tecar
Tecar Shock Wave
Shock Wave Plasma
Plasma Laser
Laser Tesla
Tesla Nikola
Nikola Kayser
Kayser Fusion
Fusion